Our research activity is focused on the development of new stereoselective synthesis methods based on the reactivity of metal enolates and their application to the synthesis of biologically active compounds.
Therefore, our work focuses on the following three fields or areas of research:
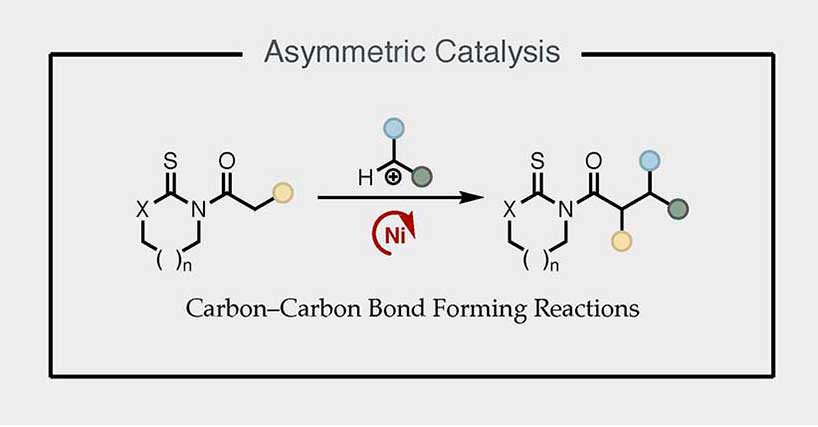
Asymmetric Catalysis
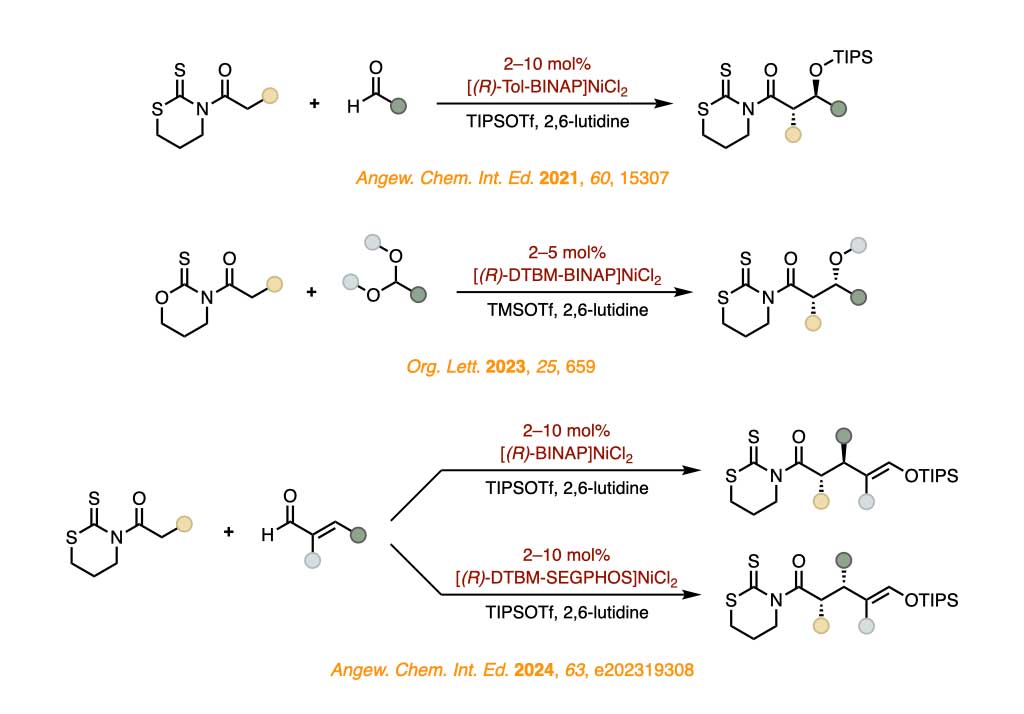
Our group focuses on the development of new asymmetric carbon–carbon bond forming reactions from thioimides, which include alkylation, aldol, Michael, and Mannich reactions as well as pericyclic cycloadditions, catalyzed by robust nickel(II) or cobalt(II) chiral complexes, thereby enabling stereodivergent strategies that provide enantiocontrolled access to all resultant stereoisomers.
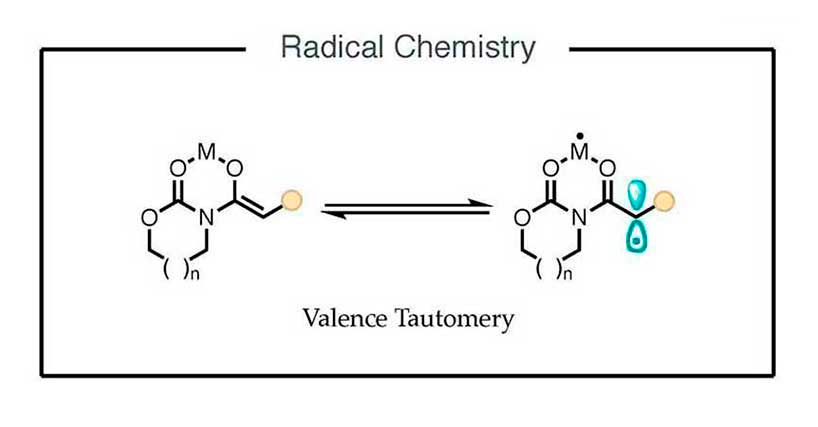
Radical Chemistry
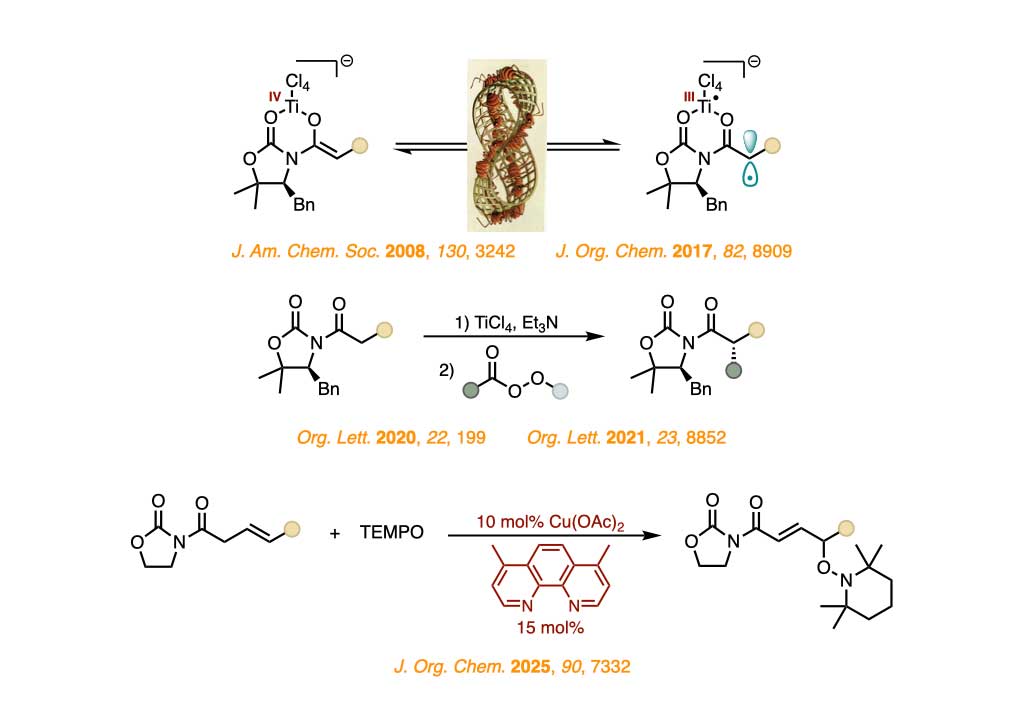
The discovery of the biradical character of titanium(IV) enolates gave rise to a new research area centered on understanding this unexpected valence tautomerism and harnessing the resultant radical-like reactivity.
We currently strive for translating this key insight into new
transformations catalyzed by metal complexes to establishing new approaches to the construction of carbon–oxygen and carbon–carbon bonds.
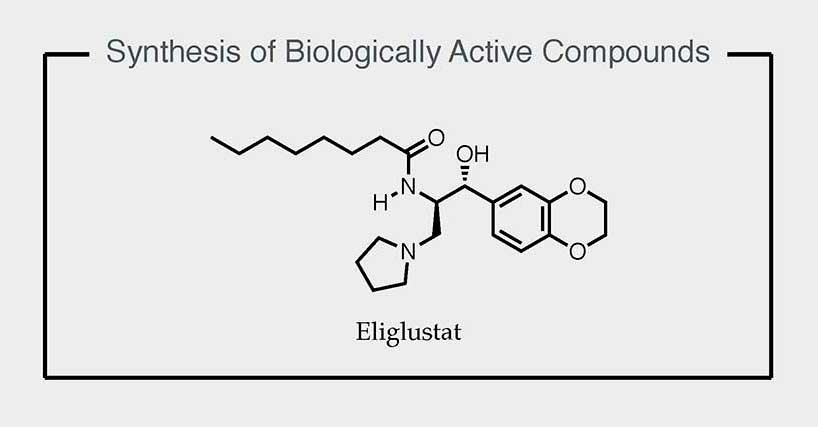
Synthesis
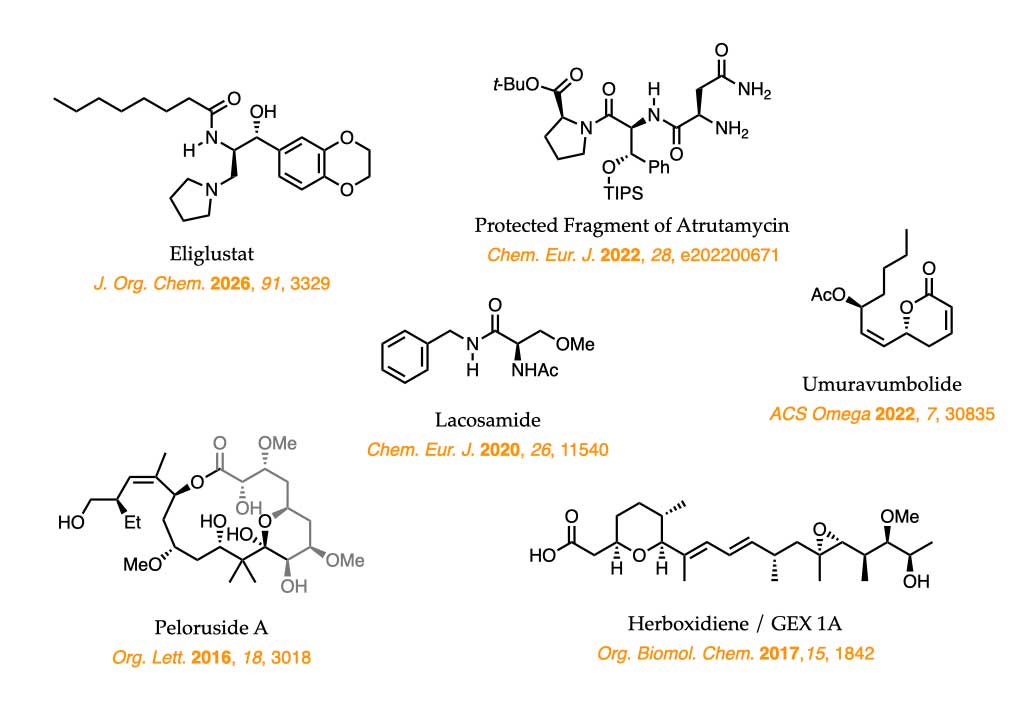
The methodologies developed in our group are applied to the
synthesis of biologically active compounds, which serve both as stringent benchmarks for assessing their suitability and as a source of inspiration for the continued development of new, simple, and effective asymmetric transformations.
If interested in joining our research group
For Master students, PhD students and Postdoctoral Researchers.